Hoheria lyallii
Sponsor
Kindly sponsored by
a member of the International Dendrology Society
Credits
Julian Sutton (2021)
Recommended citation
Sutton, J. (2021), 'Hoheria lyallii' from the website Trees and Shrubs Online (treesandshrubsonline.
Genus
Common Names
- Mountain Ribbonwood
Synonyms
- Gaya lyallii (Hook. f.) Baker f.
- Gaya lyallii var. ribifolia T. Kirk
- Plagianthus lyallii (Hook. f.) Benth.
- Plagianthus ribifolius Allan
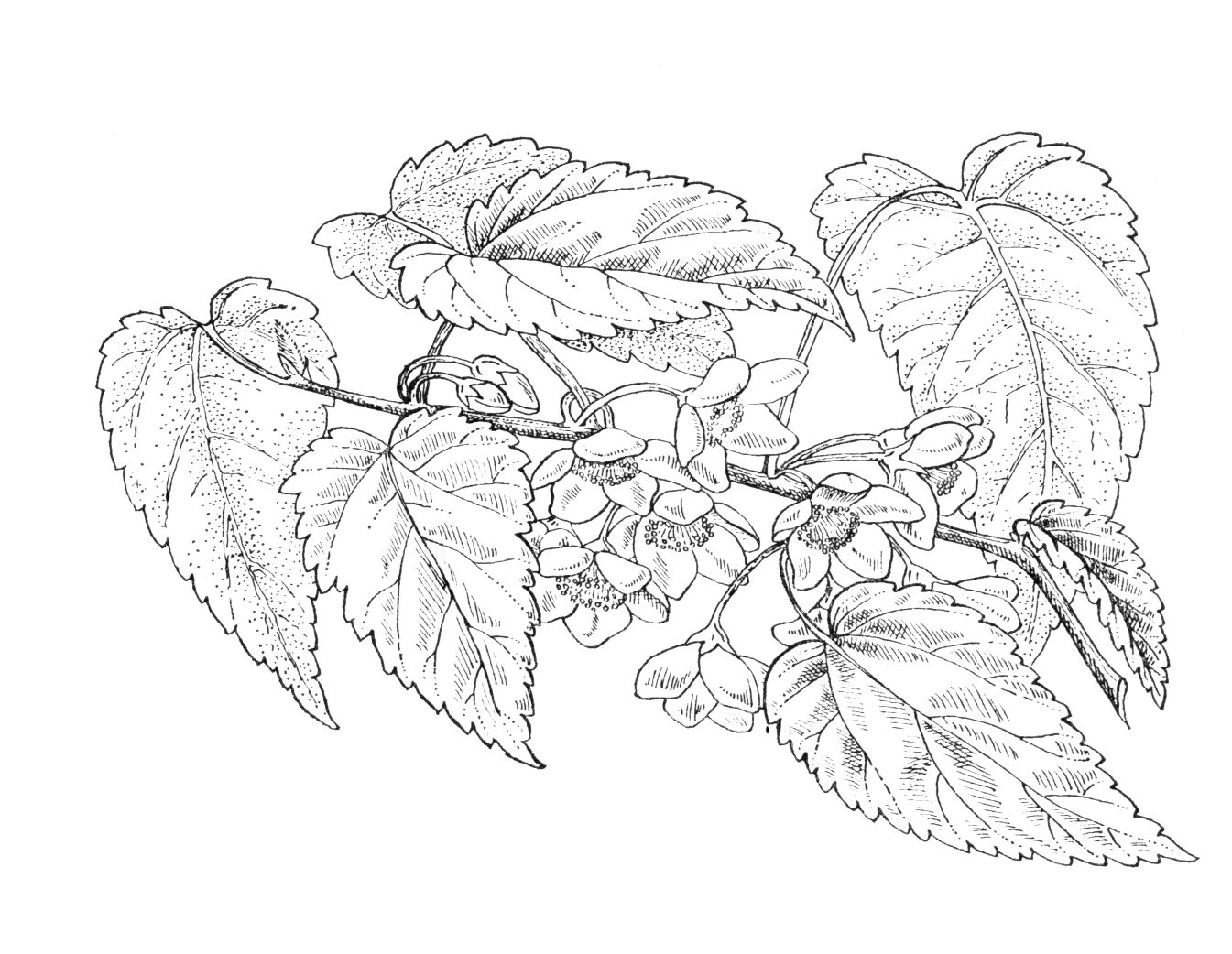
Small, deciduous tree to 8 m, heteroblastic in leaf but not growth form. Leaves of adult plant: blade elliptic to deltoid, 3.3–15.6 × 2.2–8.9 cm, both surfaces with moderate to dense stellate hairs; weakly truncate, sometimes subcordate or cordate; apex acute to subacute; margins crenate or double-crenate, often deeply lobed; petiole 10–43 mm, with moderate to dense stellate hairs. Juvenile leaves: blade broad-elliptic to suborbicular, 1.3–4 × 1.5–4 cm, both surfaces with sparse to moderate stellate hairs; base cordate to weakly truncate; apex subacute; margins deeply lobed to strongly crenate; petiole 15–52 mm, with sparse to moderate stellate hairs. Flowers 2–3(–4) cm across, axillary, solitary or in cymose fasicles of 2–3; pedicels 12–25 mm, sparsely to moderately hairy. Calyx campanulate, 3.5–6 × 7–9.5 mm, with dense stellate hairs; lobes 5–6, 3.5–6 × 3.5–5 mm, triangular to narrowly triangular, apex acute. Petals 5(–6), white, oblong-orbicular to broadly oblong, 16–20 × 11.5–15 mm, upper surface and margin towards the base with scattered simple hairs; lower surface sparsely to moderately hairy; claw 1.5–2.5 mm long. Stamens 35–50; filaments 8.5–11 mm, white, in pairs and adnate for ¼ to ¾ of their length, column with stellate and simple patent hairs; anthers 0.6–0.8 mm long. Carpels 8–14; ovary ovoid, 1–2 × 1–2 mm, densely hairy; style 8.5—10 mm, pink, sparsely hairy, fused in lower ½; stigma capitate to slightly decurrent with style. Mericarp body 5–6 × 3.5–5.5 mm, broadly elliptic, laterally compressed; wing 1—2 mm wide, extending from upper 2/3 of dorsal surface, weakly ribbed, moderately hairy, margin irregularly toothed. Seeds orange brown, glabrous, semicircular to triangular, broader toward base, sometimes with a narrow wing, usually separating from mericarp at maturity. Flowering February–March (New Zealand), July–early August (UK); Fruiting April–July (New Zealand). (Heenan et al. 2005; New Zealand Plant Conservation Network 2021)
Distribution New Zealand Eastern South Island
Habitat Open and disturbed areas, montane to subalpine.
USDA Hardiness Zone 8-10
RHS Hardiness Rating H4
Awards AGM
Conservation status Not evaluated (NE)
One of the two very similar Mountain Ribbonwood species, the deciduous Hoheria lyallii is among the earlier flowering hoherias in gardens, in Britain typically peaking in July. A rather dense covering of stellate hairs gives the leaves a greyish cast not seen in other species, while the large white flowers are quite densely clustered towards the branchlet tips among the newly emerged leaves, resembling cherry blossom in general effect. It is one of the hardier and more drought-tolerant species.
H. lyallii and H. glabrata are very similar, historically confused as wild plants and still often misidentified in gardens. As wild plants they hybridize and intergrade in a few areas where their ranges overlap. They are presumably closely related in some way, but molecular data do not confirm a simple ‘sister’ relationship; past hybridization has probably been important in Hoheria evolution (Wagstaff, Molloy & Tate 2010). The taxonomic status of the pair has been reviewed by Heenan et al. (2005), taking into account both morphological and molecular data. The key differences are their hairiness and geographical distribution. H. lyallii has a moderate to dense covering of whitish stellate hairs on both leaf surfaces and on the young branchlets; in H. glabrata they are sparse or absent. H. lyallii is found on the drier eastern flank of the Southern Alps, mainly in Marlborough and Canterbury Regions (Heenan et al. 2005). It typically occurs in scrub, in open and disturbed areas, often on nutrient-rich mineral soils. H. glabrata grows to the west of the mountains, a strikingly more rainy area. Other differences are useful, but less diagnostic. Adult leaves of H. lyallii are usually a little smaller, with truncate bases and acute tips, while those of H. glabrata are typically cordate and acuminate (Heenan et al. 2005); these differences are not absolute. Some further distinctions made in the garden literature (Bean 1981, Hutchins 2006) are perhaps based on study of too limited a range of material. While both species have a distinct juvenile leaf form, neither has the densely bushy ‘divaricating’ juvenile plant form seen in some other hoherias.
This species was described scientifically in 1853, from material collected on the Antarctic voyage of the British discovery ships Erebus and Terror; the specific epithet commemorates Scottish botanist David Lyall, assistant surgeon on HMS Terror. Hooker’s description makes it clear that he was aware of a less hairy western form ‘possibly belonging to a different species’ (Hooker 1853). However, H. glabrata remained unnamed until 1926, by which time material of both species was in British cultivation, all labelled H. lyallii. The confusion persists. Bean (1981) felt that H. glabrata was probably the commoner plant, regardless of the label, and this is perhaps still true. These species set copious seed in gardens, and there seem to be few barriers to breeding in the genus, so hybridization between them and with other species has certainly further muddied the water. Graham Hutchins (2006) also noted the evergreen, glabrous-leaved H. populnea labelled as H. lyallii in Britain. Hutchins, a British nurseryman, introduced two H. lyallii cultivars selected from wild origin seed in the late 20th century (see below). Colin Rogers claims never to have seen cultivated British material looking as grey in leaf as wild plants in New Zealand (pers. comm. 2021). While this might reflect genetic contamination of stocks, it may instead be a result of the less intense light at higher latitudes.
As with H. glabrata, most records of cultivation outside New Zealand are from the British Isles. Quite when genuine H. lyallii reached Britain is unclear, but it was probably grown by the early decades of the 20th century. Although quite hardy and drought tolerant (Hutchins (2006) grew it well in Essex) most records of larger specimens are from the wetter western fringes of Britain, and from coastal Ireland. Examples include trees at Lukesland, Devon (11 m × 53 cm in 2015) and at Mount Usher, Co. Wicklow, Ireland (16 m × 144 cm in 2015); the Tree Register (2021) wisely treats these identifications with caution, especially since it may be impossible for a visitor to reach the leaves of a taller tree to examine hairs. Aware of the difference, Bean (1981) mentions a fine specimen at Wakehurst Place, Sussex, a drier part of England.
This species would probably suit the western and northern fringes of France, and parts of the Low Countries. It is recorded from Ghent Botanical Garden, Belgium (Plantcol 2021). Hoherias in general are scarcely grown in North America. H. lyallii is recorded at Washington Park, Seattle (University of Washington Botanic Gardens 2021), and would seem a good bet for the Pacific Northwest, west of the Cascades.
'Chalk Hills'
Leaf margins crenate; two deep incisions almost make the leaf 3-lobed. A clonal cultivar selected from seed collected from a tree at the base of a scree at about 1000 m near Swale Stream in the Chalk Range, Marlborough Region, by Graham Hutchins in 1985. Distributed from cuttings by Hutchins’ County Park Nursery, Essex (Hutchins 2006).
'Swale Stream'
A sister seedling to ‘Chalk Hills’ with irregularly crenate leaves, the crenations more shallow than in ‘Chalk Hills’, and without deeper incisions suggesting lobing (Hutchins 2006).